MANA RBM's network of clinical researchers reported that research sites are eliminating onsite monitoring visits. If onsite monitoring visits are a Sponsor's main oversight approach, the solution to this critical problem is available now.
MANA RBM provides remote, automated risk-based monitoring (RBM) customized to a site's protocol. The company delivers its solution, customized to the areas of greatest concern, specific to a study. Subject review can begin within days.
"We recently published an example of this successful approach, adding remote Risk Based Monitoring to trials being conducted with traditional SDV," said Penny Manasco, M.D., CEO of MANA RBM. "Within days, we started remote subject review. Within 2 months, we identified critical errors that SDV did not find."
Here is the link to the head to head comparison:
http://www.appliedclinicaltrialsonline.com/comparing-risk-based-monitoring-and-remote-trial-management-vs-sdv
March 17, 2020
Topic Area: Press Release
Recent Posts
 Building Senior Living for the Next Generation of Retirees
Building Senior Living for the Next Generation of Retirees
As baby boomers seek new retirement expectations, developers and designers must rethink senior living facilities
 Managing Drain and Biofilm Risk
Managing Drain and Biofilm Risk
Drains are out of sight, but a coordinated program linking infection prevention, EVS and facilities can keep them from getting out of control.
 Froedtert Hospital Blue Parking Facility IPMI Earns Award for Excellence
Froedtert Hospital Blue Parking Facility IPMI Earns Award for Excellence
It is one of the parking and mobility industry’s most prestigious global honors.
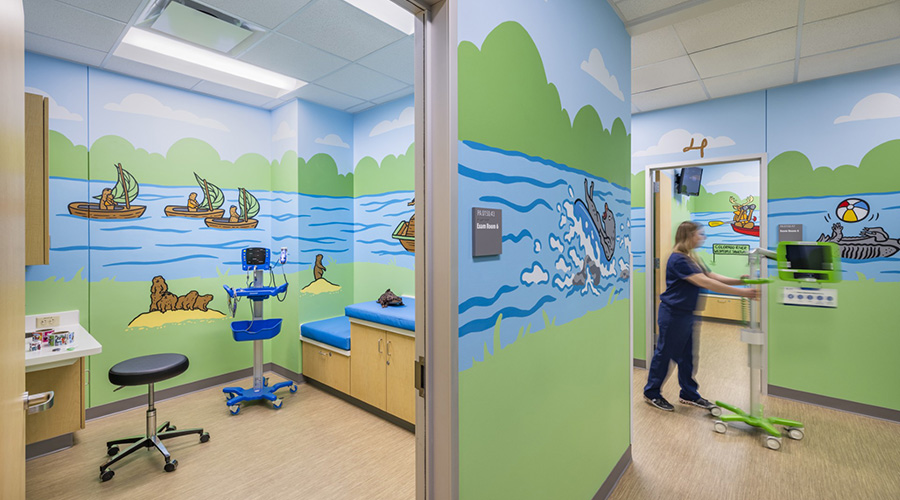 The Role of Positive Distraction in Pediatric Design
The Role of Positive Distraction in Pediatric Design
Positive distraction by itself does not heal, but it can aid the healing process by addressing the mental well-being of an individual.
 Healthcare Waste is Fueling America's Debt
Healthcare Waste is Fueling America's Debt
As healthcare spending surpasses $5 trillion annually, facility leaders are under pressure to confront operational inefficiencies head-on.