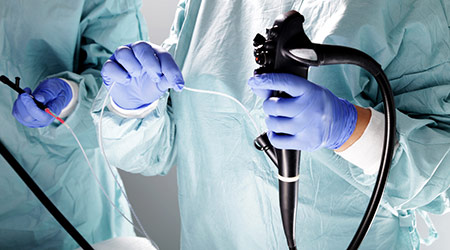
Two recent studies suggest that current reprocessing methods do not effectively sterilize flexible endoscopes, according to an article on the Becker's Clinical Leadership & Infection Control website.
In a study in the American Journal of Infection Control, researchers found that 22 percent of endoscopes had signs of organic contamination and 71 percent still contained microbial growth after reprocessing.
The researchers took samples of fully reprocessed endoscopes stored for at least 24 hours.
Researchers in the Netherlands found at least one reprocessed duodenoscope contaminated with detectable microbes in 39 percent of all Dutch healthcare facilities studied.
 Making Healthcare Lighting Retrofits Work
Making Healthcare Lighting Retrofits Work Stadium Design is Reshaping Healthcare Facilities
Stadium Design is Reshaping Healthcare Facilities AHN Reveals Plans to Build New Canonsburg Hospital in Pennsylvania
AHN Reveals Plans to Build New Canonsburg Hospital in Pennsylvania Designing for Distraction: Benefits for Children, Families
Designing for Distraction: Benefits for Children, Families Staffing and Consolidation Reshape Outpatient Facility Strategies
Staffing and Consolidation Reshape Outpatient Facility Strategies