Preliminary results from the US Food and Drug Administration's (FDA's) mandated postmarket surveillance studies of duodenoscopes show "higher-than-expected" contamination rates after reprocessing, according to an article on the Medscape website.
The FDA said 3 percent of properly collected samples tested positive for more than 100 colony-forming units of "low-concern" organisms.
These organism are unlikely to cause serious infections but are, nevertheless, an indication of a "reprocessing failure."
An additional 3 percent of properly collected samples tested positive for "high-concern" bacteria that are more often associated with disease.
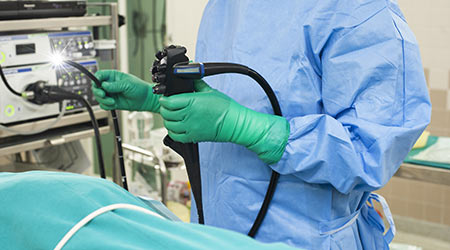
 A Cleaning Alternative: The Benefits of Steam Technology
A Cleaning Alternative: The Benefits of Steam Technology Novant Health Gets Approval for New Hospital and Freestanding Emergency Department
Novant Health Gets Approval for New Hospital and Freestanding Emergency Department Preparing Healthcare Facilities for Severe Thunderstorms
Preparing Healthcare Facilities for Severe Thunderstorms NLCS Strengthens Safety and Compliance with Comprehensive Electrical Program
NLCS Strengthens Safety and Compliance with Comprehensive Electrical Program University of South Carolina Opens New Brain Health Center
University of South Carolina Opens New Brain Health Center