A new ASTM International standard describes the use of pigskin as an alternative to human hands in testing the efficacy of hand hygiene products in combating contamination.
ASTM E2897, Guide for Evaluation of the Effectiveness of Hand Hygiene Topical Antimicrobial Products Using Ex Vivo Porcine Skin, overcomes the limits in the types of pathogens that can be used in human testing, says ASTM. The standard was approved by Committee E35 on Pesticides, Antimicrobials and Alternative Control Agents and developed by Subcommittee E35.15 on Antimicrobial Agents.
As well, while testing hygiene products on human hands can be done, tests involving human volunteers can be expensive.
“ASTM E2897 will permit testing of pathogens that may be encountered in a healthcare environment but would not be tested on human subjects,” says Peter Karanja, research and development scientist, STERIS Corp., and an E35 member. “It also permits testing of conditions that cannot be simulated in a test tube while offering less expensive and higher throughput screening hand hygiene products.”
In addition to ASTM E2897, E35.15 is developing a related proposed standard, ASTM WK36911, Guide for Measuring the Inactivation of Persistent Activity of Topical Antimicrobial Products Using Ex Vivo Porcine Skin. This proposed guide will demonstrate the compatibility of topical antimicrobial products and the ability to maintain persistent activity exhibited by products used in a healthcare setting. Like ASTM E2897, ASTM WK36911 uses pigskin as a surrogate for human skin.

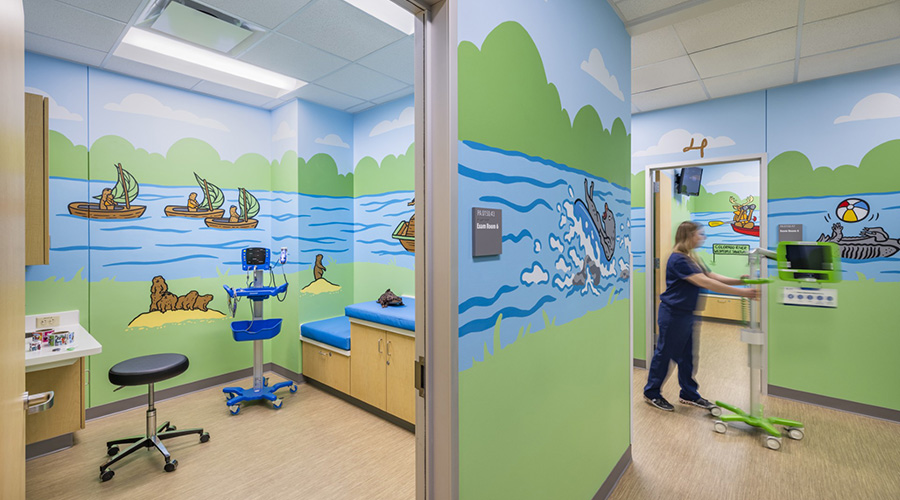 The Role of Positive Distraction in Pediatric Design
The Role of Positive Distraction in Pediatric Design Healthcare Waste is Fueling America's Debt
Healthcare Waste is Fueling America's Debt Prairie Lakes Healthcare System to Rebrand Following Sanford Health Merger
Prairie Lakes Healthcare System to Rebrand Following Sanford Health Merger How Digital Technologies Are Reshaping Performance in Healthcare Facilities
How Digital Technologies Are Reshaping Performance in Healthcare Facilities The Role of Plumbing in Healthcare-Associated Infections
The Role of Plumbing in Healthcare-Associated Infections