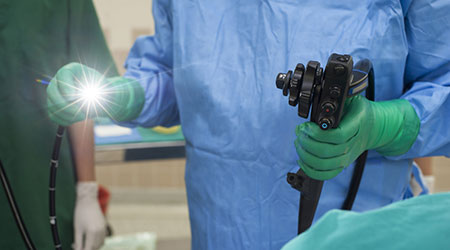
FDA recently granted Pentax approval to market a duodenoscope with fewer parts that need to be cleaned, according to an article on the Advisory Board website.
According to FDA, as many as 350 patients at 41 medical facilities across the globe were infected by or exposed to contaminated gastrointestinal scopes between 2010 and 2015.
Contaminated scopes had killed at least 35 patients in the United States from 2013 to January 2017.
FDA in 2013 started to collaborate with duodenoscope manufacturers Fujifilm, Olympus, and Pentax to address contamination risks.
 Designing for Distraction: Benefits for Children, Families
Designing for Distraction: Benefits for Children, Families Staffing and Consolidation Reshape Outpatient Facility Strategies
Staffing and Consolidation Reshape Outpatient Facility Strategies Adams Health Network Falls Victim to Phishing Attack
Adams Health Network Falls Victim to Phishing Attack Ventilation and Filtering for Infection Control
Ventilation and Filtering for Infection Control  ChristianaCare Opens Aston Campus Neighborhood Hospital
ChristianaCare Opens Aston Campus Neighborhood Hospital