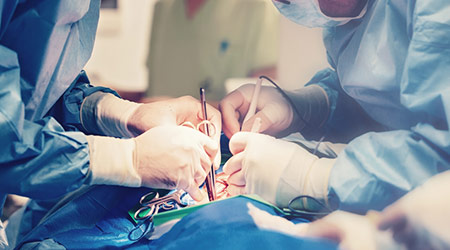
The US Food and Drug Administration (FDA) has issued guidance on preventing surgical fires, according to an article on the Medscape website.
The FDA recommends the safe use of medical devices and products commonly used during surgical procedures.
To reduce the risk for surgical fires, the FDA suggests healthcare providers and hospital staff who perform surgical procedures should be educated on the factors that increase the risk for surgical fires, as well as how to manage fires.
The FDA also recommends that staff perform a fire risk assessment at the beginning of each surgical procedure.
 Designing for Distraction: Benefits for Children, Families
Designing for Distraction: Benefits for Children, Families Staffing and Consolidation Reshape Outpatient Facility Strategies
Staffing and Consolidation Reshape Outpatient Facility Strategies Adams Health Network Falls Victim to Phishing Attack
Adams Health Network Falls Victim to Phishing Attack Ventilation and Filtering for Infection Control
Ventilation and Filtering for Infection Control  ChristianaCare Opens Aston Campus Neighborhood Hospital
ChristianaCare Opens Aston Campus Neighborhood Hospital