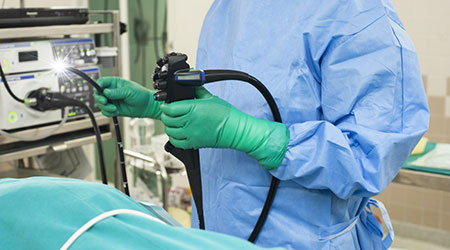
The Food and Drug Administration (FDA) said companies that make duodenoscopes should begin making disposable versions, according to an article on The New York Times website.
The agency also said that hospitals should start to transition to models with disposable components to reduce the risk of infection.
FDA tests revealed that one in 20 harbored disease-causing microbes like E. coli even after being properly cleaned.
Medical experts have urged the FDA to force manufacturers to make the scopes safer and easier to decontaminate — or to take them off the market.
 Making Healthcare Lighting Retrofits Work
Making Healthcare Lighting Retrofits Work Stadium Design is Reshaping Healthcare Facilities
Stadium Design is Reshaping Healthcare Facilities AHN Reveals Plans to Build New Canonsburg Hospital in Pennsylvania
AHN Reveals Plans to Build New Canonsburg Hospital in Pennsylvania Designing for Distraction: Benefits for Children, Families
Designing for Distraction: Benefits for Children, Families Staffing and Consolidation Reshape Outpatient Facility Strategies
Staffing and Consolidation Reshape Outpatient Facility Strategies