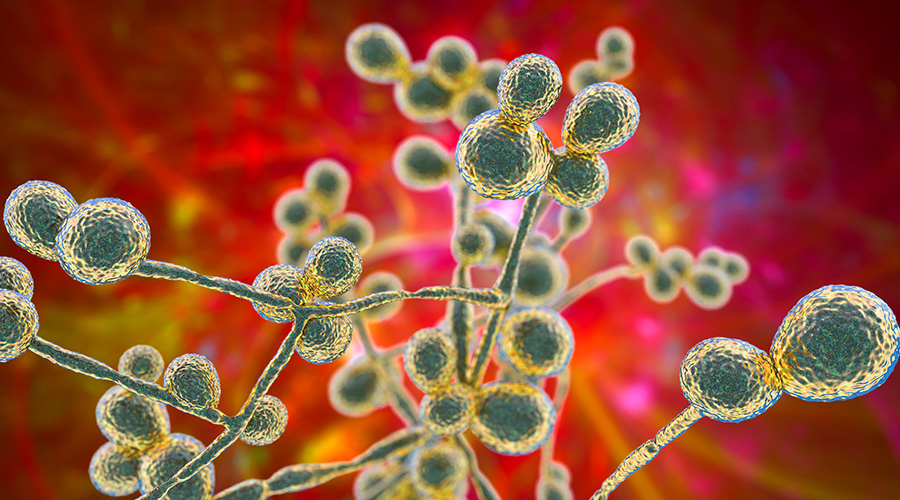
No standardized, validated, real-world test method exists for evaluating the performance of hospital-grade disinfectants against dry surface biofilms. That makes it extremely difficult to know whether a hospital disinfectant can truly kill Candida auris (C. Auris) or other multi-drug resistant organisms (MDRO) once they are embedded in these mature structures.
Biofilms frequently are associated with healthcare-associated infections. To reduce patient risks, a hospital-approved disinfectant should be able to inactivate bacteria in biofilms. Multiple methods are described in the literature, but no consensus has been reached for disinfection efficacy tests against biofilms. This study demonstrates a direct relationship between biofilm age — maturation time — and biofilm resistance. Specifically:
- Longer-grown biofilms are more mature and more robust. ISO 15883-5 biofilms grown for 96 hours contained higher levels of protein, total organic carbon (TOC) and bacterial load than ASTM E2652 biofilms grown for 48 hours and Konrat et al. biofilms grown for 24 hours.
- Mature biofilms are significantly harder to kill. Disinfection efficacy tests showed that the longer the biofilm maturation phase, the lower the disinfectant’s effectiveness.
- As for regulatory and type-test implications, disinfectants intended for use on medical devices or healthcare surfaces should be tested against mature biofilms, not against young or early-stage biofilms, because immature biofilms underestimate real-world resistance.
Short-growth biofilm models underestimate risk. Biofilms grown for 24-48 hours are not representative of the mature, clinically relevant biofilms found on medical devices or dry hospital surfaces.
Mature biofilms behave like a different organism. Their extracellular matrix is denser, their metabolic activity is lower, their chemical penetration is reduced, and their tolerance to disinfectants is dramatically higher.
Related Content: Strategies to Eradicate Biofilm Containing C. Auris
This mirrors what we see with C. auris and MDRO dry surface biofilms.
Biofilms behave nothing like chemical assays. Disinfectant testing is fundamentally a bioassay, not a chemical assay. Microbes in a biofilm have genotype- and phenotype-dependent tolerance, and this tolerance varies from carrier to carrier, even under controlled lab conditions. This biological variability makes it hard to produce consistent, reproducible results.
No standardized test method exists for dry surface biofilms. Regulatory and standards bodies — the U.S. Environmental Protection Agency (EPA), AOAC International, ASTM, ISO — have no unified method for testing disinfectants against dry surface biofilms. Existing guidance documents were written for chemical assays, not for microbial communities embedded in protective matrices like mature biofilm, which can be found on nearly every surface in the near patient zone. As a result, many critical biofilm-specific factors are simply not addressed.
Dry surface biofilms are structurally complex and highly variable. They are mixed-species, low-moisture, slow-metabolism communities that form on plastics, metals, wood, textiles and other hospital materials. Their composition and architecture vary, making it difficult to create a standard biofilm for testing.
This means no two dry surface biofilms are identical, kill claims may depend on the exact biofilm grown, and results might not translate to real hospital surfaces.
No EPA-approved method exists for testing disinfectants against C. auris in dry surface biofilm. The EPA has no validated, regulatory test method for:
- mixed-species biofilms
- dry surface biofilms
- biofilm-embedded C. auris
- disinfectant penetration through extracellular matrix.
This is why disinfectants on EPA’s List P cannot be assumed to kill C. auris in a dry surface biofilm.
J. Darrel Hicks, BA, MESRE, CHESP, Certificate of Mastery in Infection Prevention, is the past president of the Healthcare Surfaces Institute. Hicks is nationally recognized as a subject matter expert in infection prevention and control as it relates to cleaning. He is the owner and principal of Safe, Clean and Disinfected. His enterprise specializes in B2B consulting, webinar presentations, seminars and facility consulting services related to cleaning and disinfection. He can be reached at darrel@darrelhicks.com, or learn more at www.darrelhicks.com.
 Cleanliness Is a Measurable Outcome
Cleanliness Is a Measurable Outcome Workplace Safety and the Role of Access Control
Workplace Safety and the Role of Access Control Henry Ford Hospital Celebrates Construction Milestone for Expansion Project
Henry Ford Hospital Celebrates Construction Milestone for Expansion Project How EVS Leaders Can Support Staff for Better Cleaning
How EVS Leaders Can Support Staff for Better Cleaning Addressing Infection Prevention Staffing Gaps in Ambulatory and Procedural Care
Addressing Infection Prevention Staffing Gaps in Ambulatory and Procedural Care